
About Us
Leveraging our streamlined operations from R & D to manufacturing, we have crafted a value-add model that effectively transitions from APIs to various formulations and new drug delivery systems.
Our outstanding 4 manufacturing facilities, stringent quality standards, robust compliance practices and an enviable regulatory track record combine to make our Finished Dosage business, a true value-based solution. Be it sterile, solid dosage forms or specialist filling, we accelerate the speed-to-market for our customers while mitigating risk.
We have initiated several strategic initiatives to strengthen and fortify our FDF business across generic, branded and contract manufacturing segments.
In a short span we expanded our MRs from 40 to a 100, achieved 30 market authorizations and ANDA’s and have set-up a contract research lab to support our own filings of MA and ANDA.
Having established ourselves with finished formulations in the semi regulated markets, we set our sights on growth in the regulated generic markets such as Europe, US, Canada & Australia.
Manufacturing Facilities
Our capabilities to meet the regulatory requirements of different countries, has enabled us to develop a rich and diverse product portfolio of branded formulations in ophthalmic, nutraceutical & dermatology. Thus, we ensure that we meet customers’ requirements in India, Southeast Asia, Africa and Middle East.
We are making a conscious effort to expand our global footprint by having alliances with global players for collaborative research, marketing tie-ups and joint ventures to create synergies.
Our 4 manufacturing facilities all of which are WHO and CGMP certified, are equipped to deliver a range of dosage delivery formats that include Capsules, Tablets, Syrups & liquids, Sprays & ointments, making us the preferred manufacturing partner for global pharmaceutical organizations.

Jeedimetla, Hyderabad (Unit 1)
- Manufactures dosage forms across delivery platforms such as liquid orals, tablets, capsules and ointments in variours therapeutic categories
- Approved by PICS nations

Jeedimetla, Hyderabad (Unit 2)
- Manufactures dosage forms across delivery platforms
- Batch compounding capacities from 100 ltrs to 500 ltrs with 3 piece HDPE/Vial filling capacities
- ISO 13485 certified
- CE certificate for medical devices

Kashipur, Uttarkhand
- Located in the area which enjoys various tax concessions
- Engaged in manufacturing non-sterile syrups, tablets, capsules, external creams and lotions
- WHOGMP certified
- ISO 9001-2000, ISO 14001 and OHSAS 18001 certifications

Haridwar
- Commenced operations in 2005 & today manufactures nasal drops, eye drops and ear drops under this facility in North India. Key customers include Novartis, Merck, Cipla, Lupin etc.
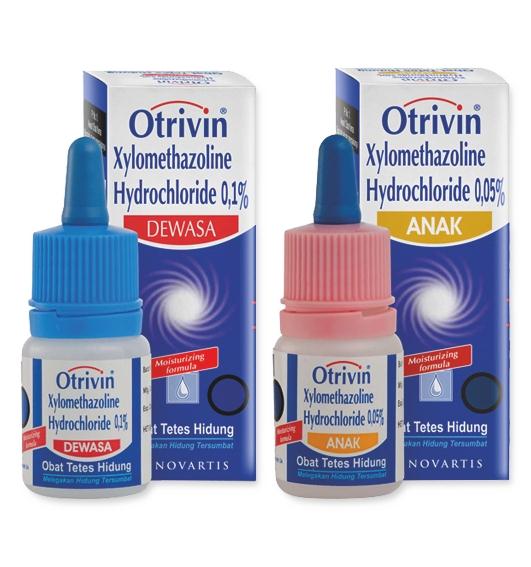
- Otrivin (for Novartis) and Nasivion (for Merck ) are the two major products being manufactured and sold at this facility
- Accreditations: WHO GMP (For ROW markets, Institutional sales); In process to procure Russia GMP and Uzbekistan GMP
Contract Manufacturing (CMO)
Vivimed serves various reputed companies through the CMO division and has been able to generate various products catering to India, Canada and ROW markets.
Capsules and Tablets: Flexasur; Spasmocip Plus; Codarin; Butaproxivon; Valenzia Tablets; Arachitol Tablet; C Pink Tablet
Syrups and liquids: Codarex; Inalgel; Viscodyne; Brozedex (sugar free); Celadrin; Mits Codeine Linctus; Candid Lotion; Candid Mouth Paint
Nasal sprays and Ointments: Otrivin; Nasivion Moist (spray); Nasivion (nasal drops); Candbiotic Ear Drop; Otrivin Nasal Spray; Tobrop
Branded Formulations
We have our branded formulations in niche and high growth therapeutic segments such as Ophthalmic, Nutraceuticals & Dermatology and Pain.
Ophthalmic: Renicol, Lysicon-V, Care Tears and Dexacort.
Nutraceuticals & Dermatology: Our range of Nutraceuticals products include enzyme preparations, B-lactams, anti-oxidants and multi-vitamins for women and children











Contact Us
India
Address: Vivimed Labs Ltd. North End, Road No.2, Banjara Hills, Hyderabad, Telangana – 500 034, India
Phone: +91(0)40 6608 6608
Fax: +91(0)40 6608 6699
Email: info@vivimedlabs.com
